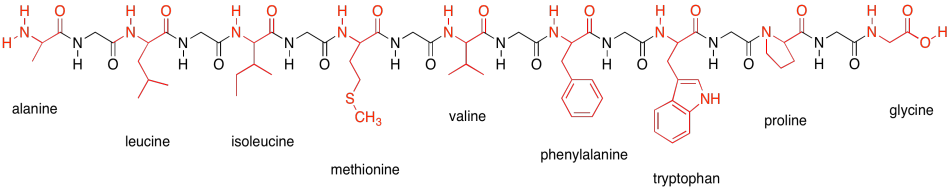
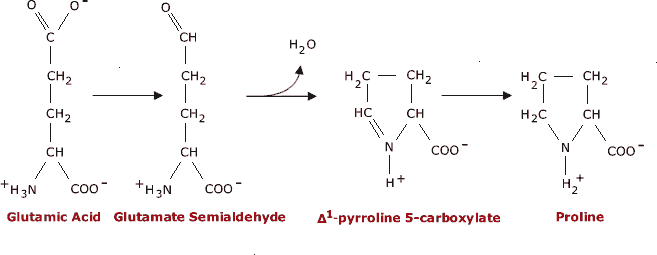
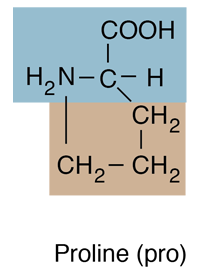
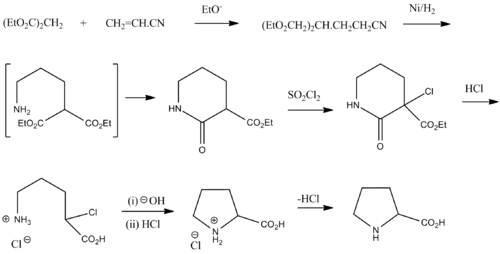
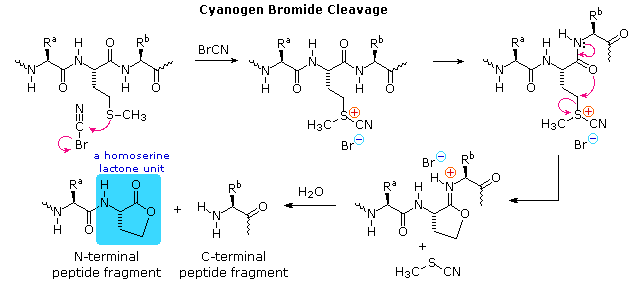
Why are glycine and proline less commonly found in alpha helices than other amino acids? | Homework.Study.com
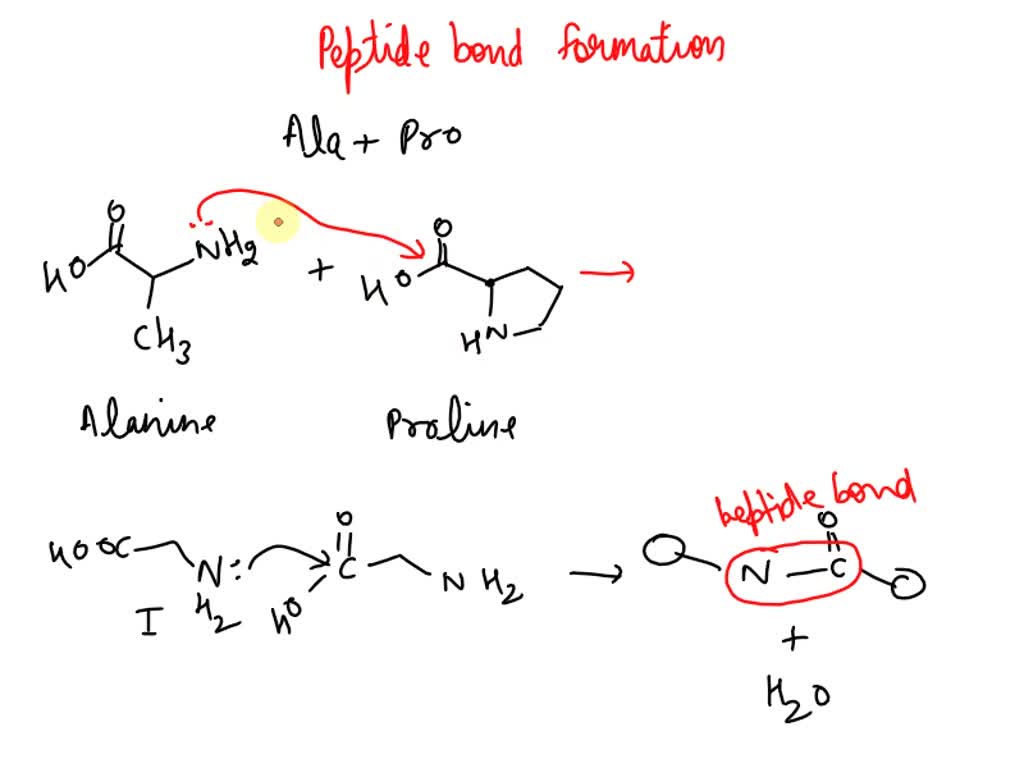
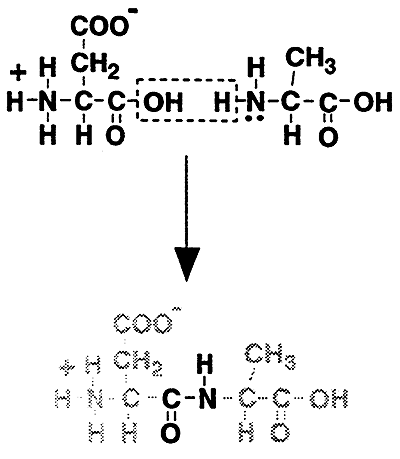
SOLVED: Draw a scheme for the reaction of the peptide bond formation between Alanine and Proline. Explain the reaction.

Evidence of the Reduced Abundance of Proline cis Conformation in Protein Poly Proline Tracts | Journal of the American Chemical Society

Main Chain Hydrogen Bond Interactions in the Binding of Proline-rich Gluten Peptides to the Celiac Disease-associated HLA-DQ2 Molecule - ScienceDirect

Enantiopure 5-CF3–Proline: Synthesis, Incorporation in Peptides, and Tuning of the Peptide Bond Geometry | Organic Letters

Probing Peptidylprolyl Bond cis/trans Status Using Distal 19F NMR Reporters - Killoran - 2023 - Chemistry – A European Journal - Wiley Online Library

Structural Basis of Proline-Proline Peptide Bond Specificity of the Metalloprotease Zmp1 Implicated in Motility of Clostridium difficile - ScienceDirect
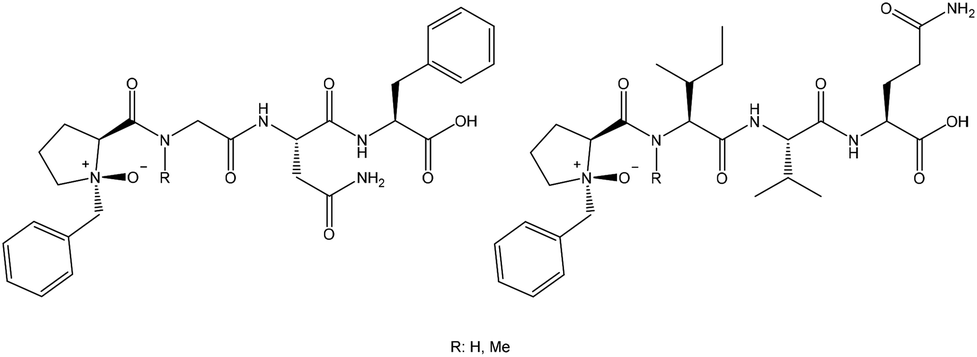
Proline N -oxides: modulators of the 3D conformation of linear peptides through “NO-turns” - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C4OB00433G
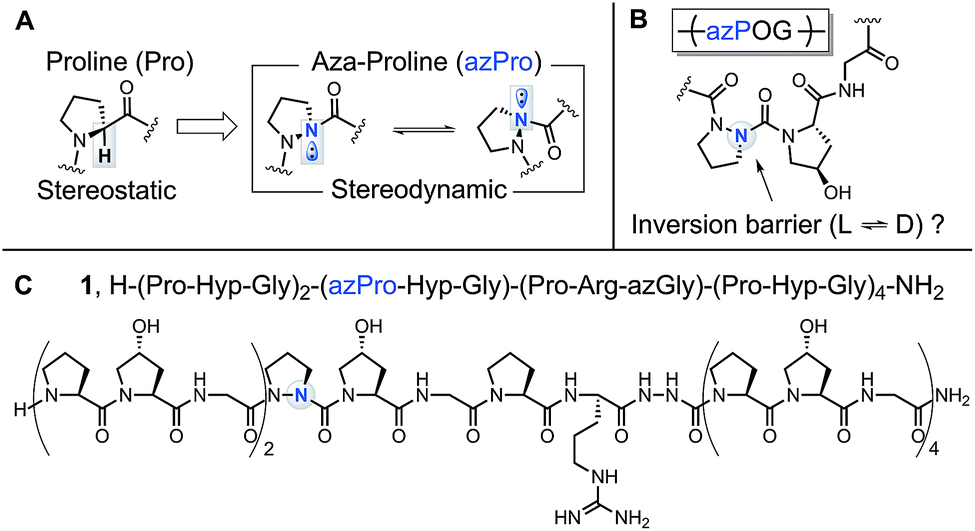
Aza-proline effectively mimics l -proline stereochemistry in triple helical collagen - Chemical Science (RSC Publishing) DOI:10.1039/C9SC02211B

The alpha-helical parts of myoglobin and other proteins stop whenever a proline residue is encountered in the chain. Why is proline never present in a protein alpha helix? | Homework.Study.com
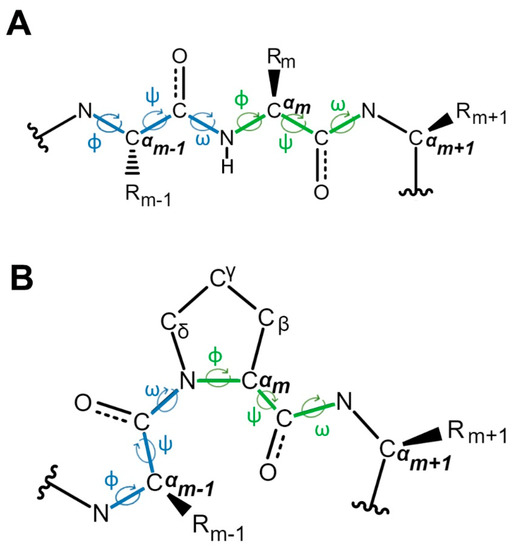
Biology | Free Full-Text | Proline Isomerization: From the Chemistry and Biology to Therapeutic Opportunities