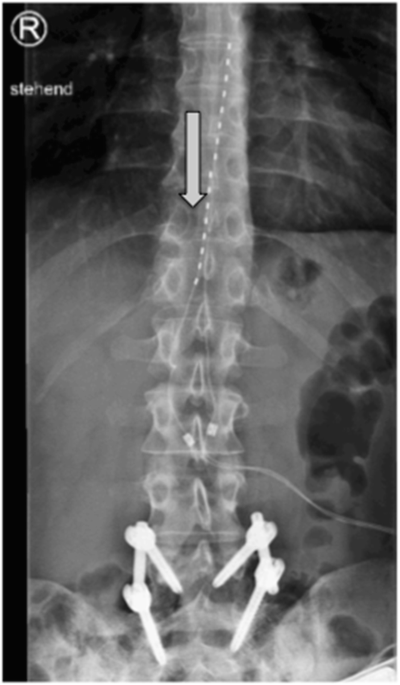
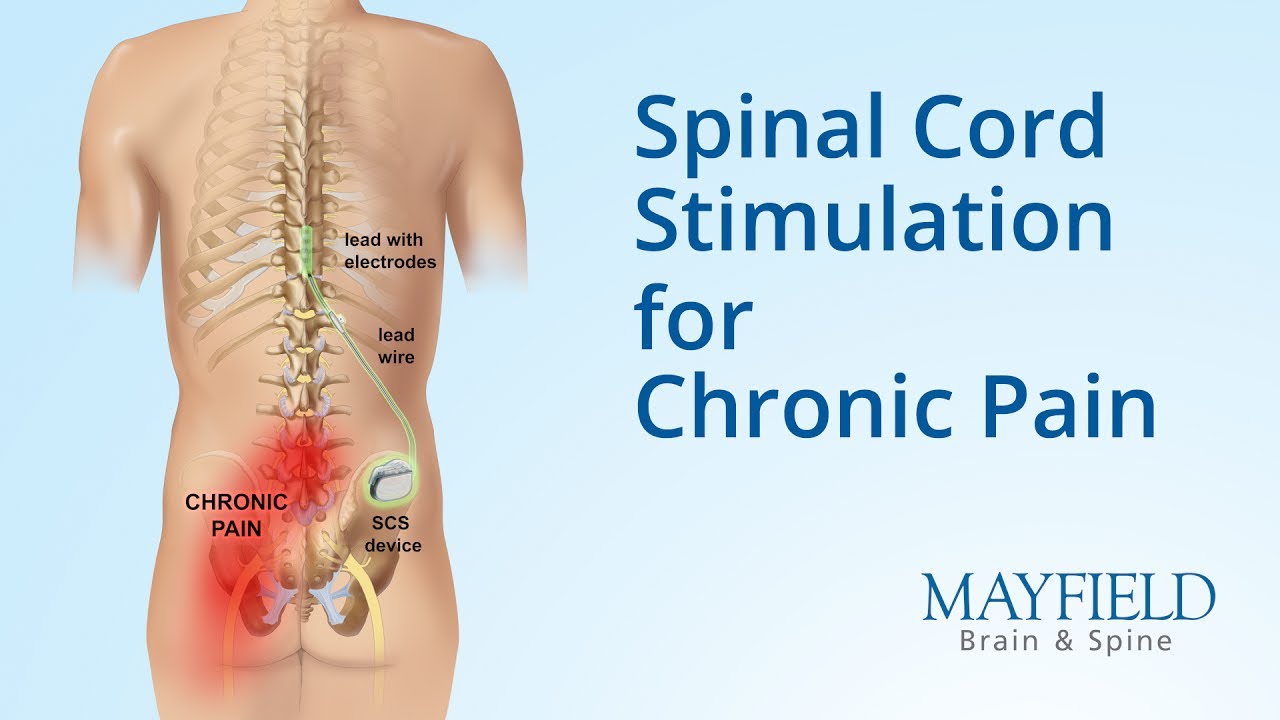
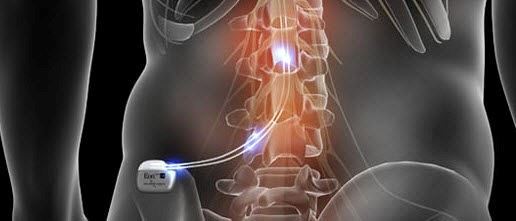
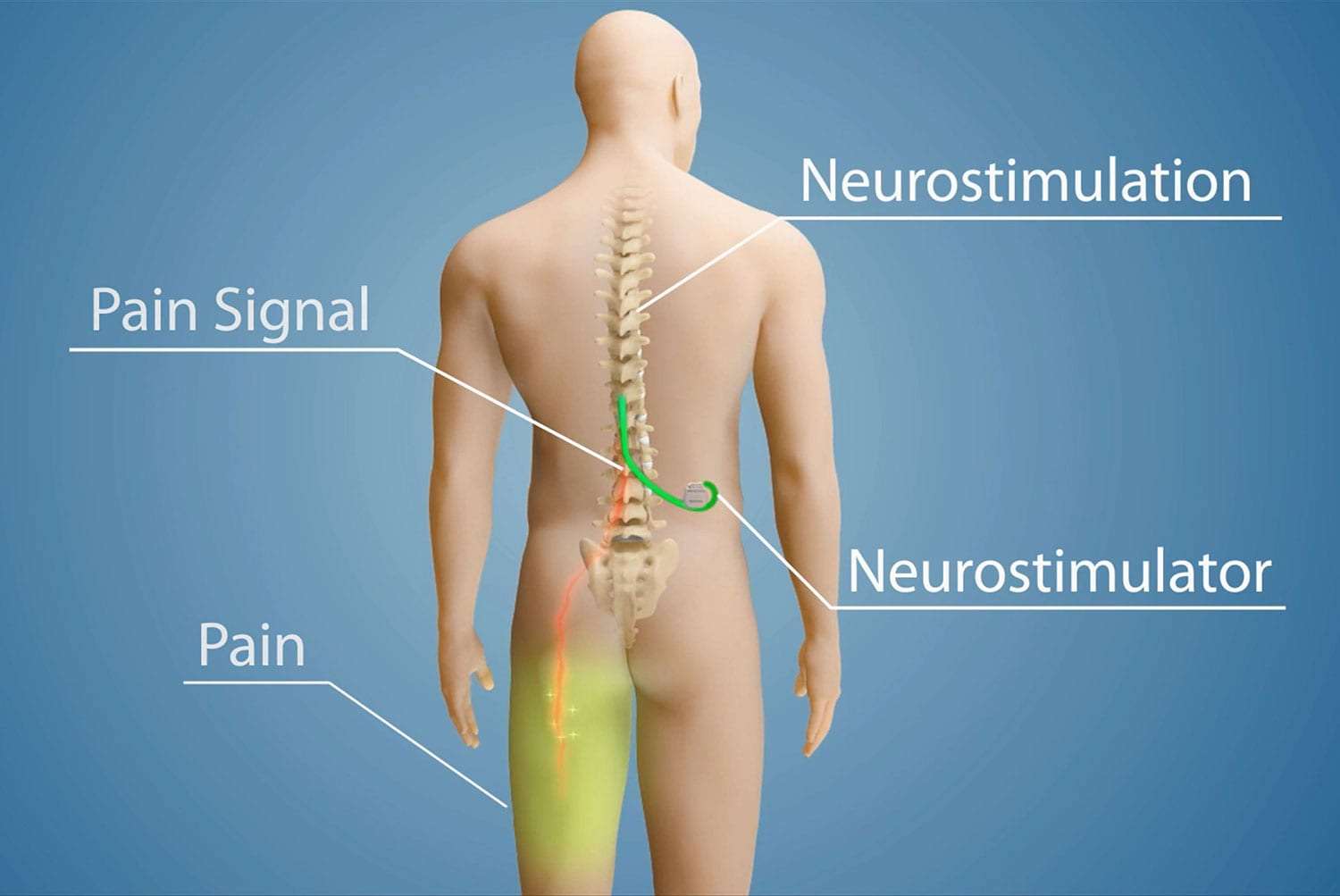
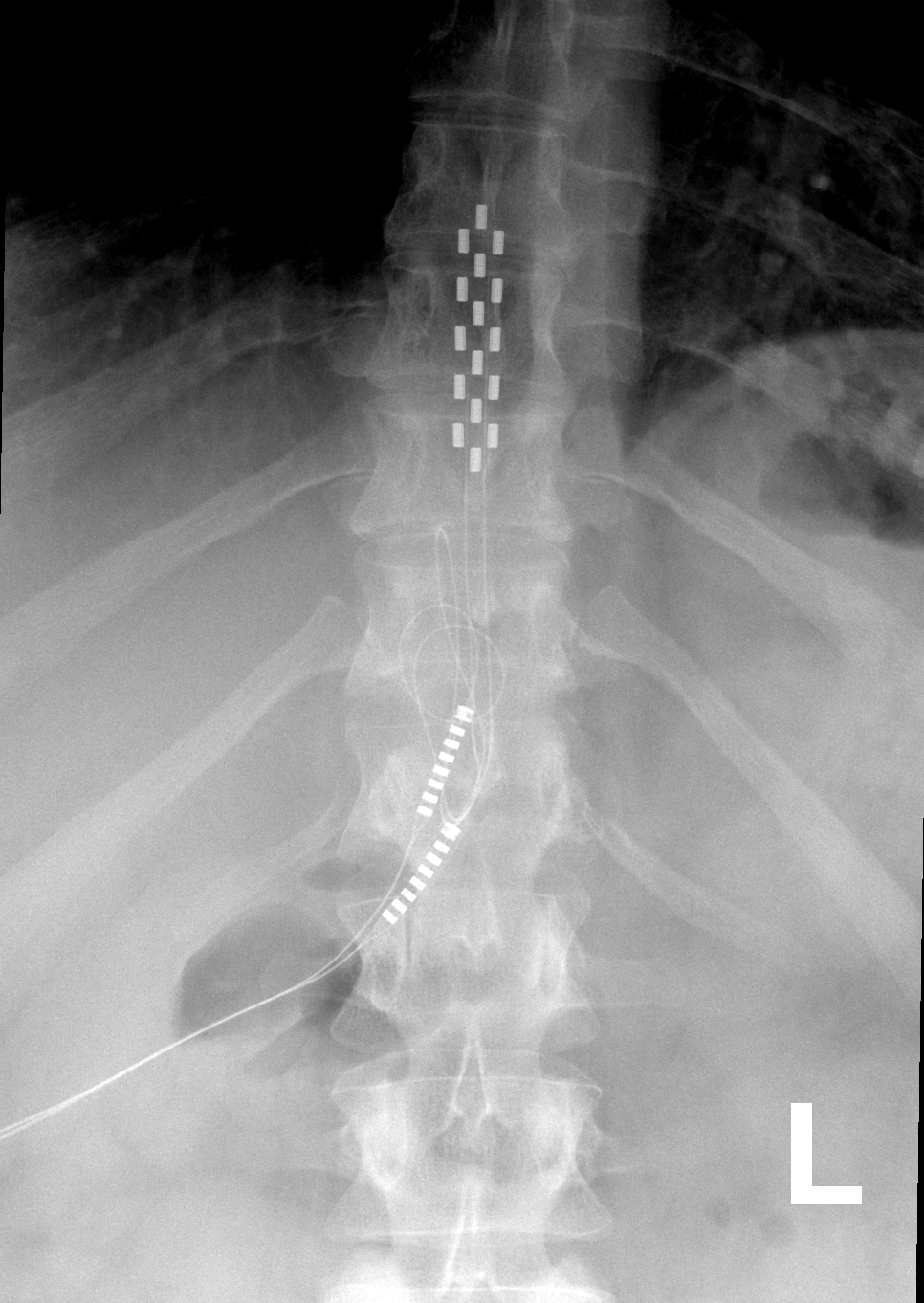
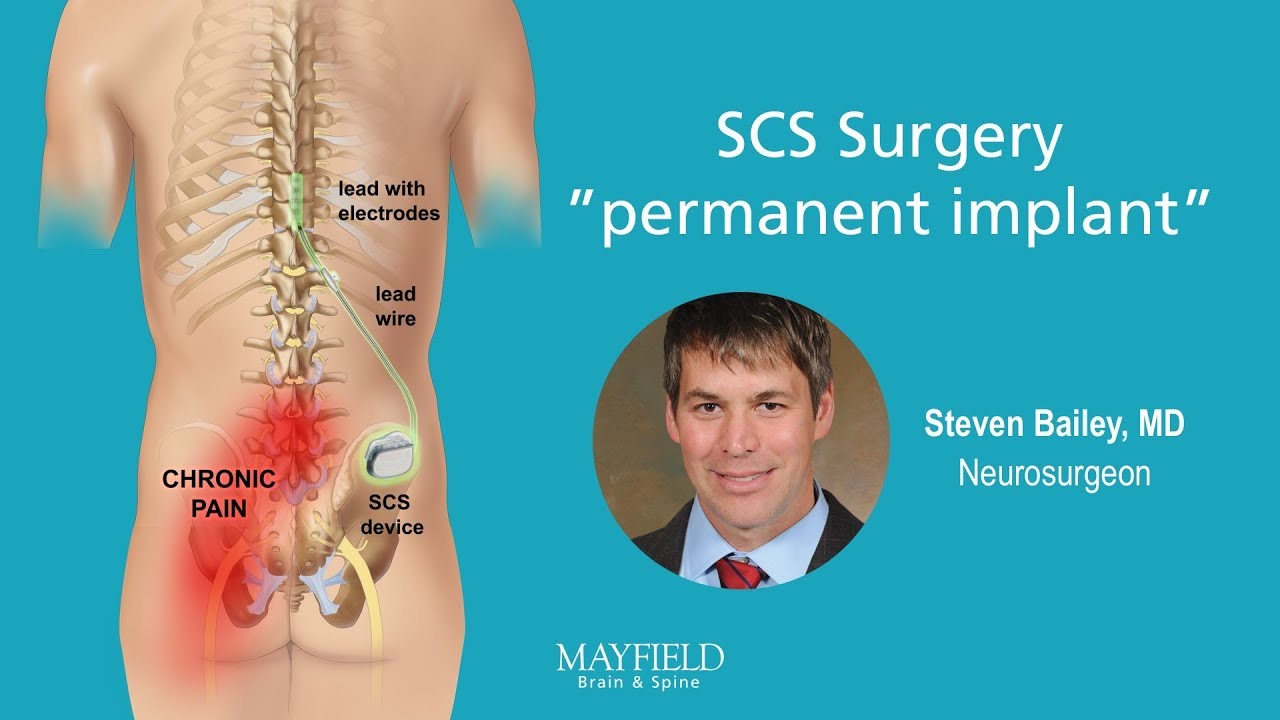
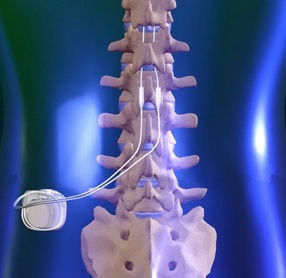
Multicentre, double-blind, randomised, sham-controlled trial of 10 khz high-frequency spinal cord stimulation for chronic neuropathic low back pain (MODULATE-LBP): a trial protocol. - Abstract - Europe PMC

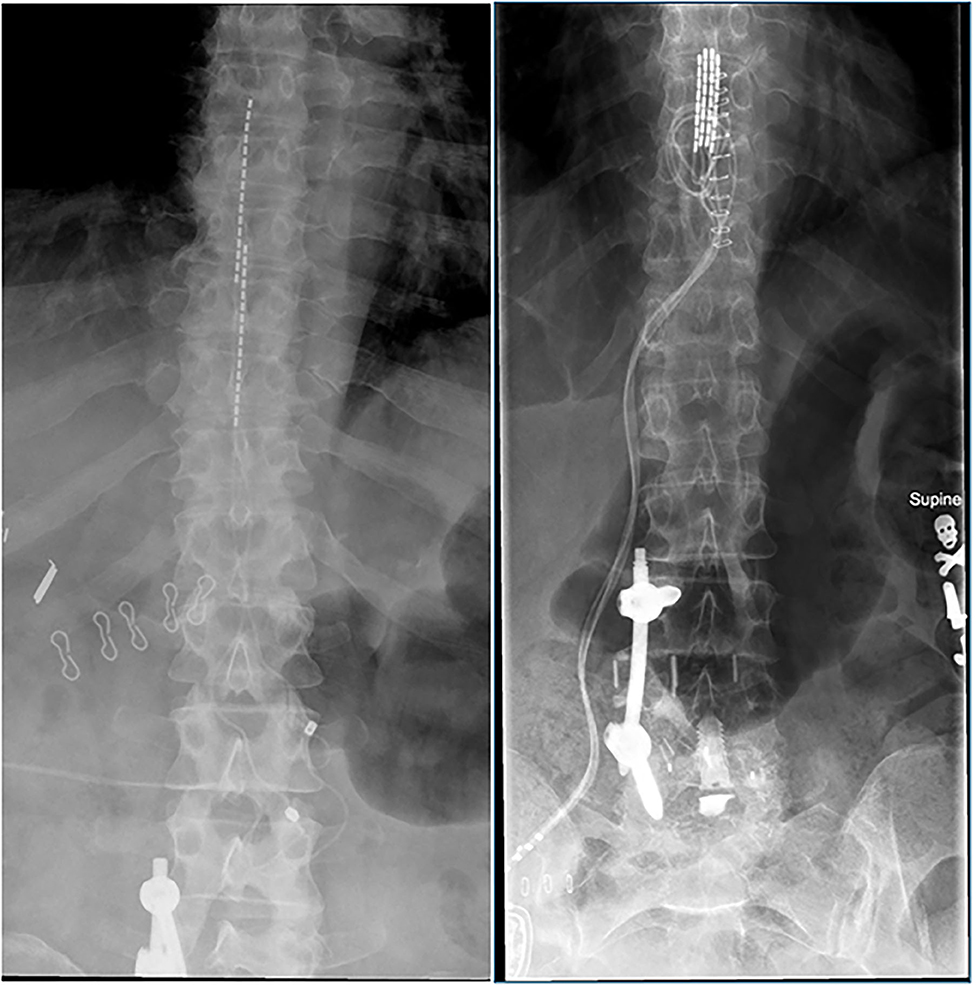
Comparison of conventional, burst and high-frequency spinal cord stimulation on pain relief in refractory failed back surgery syndrome patients: study protocol for a prospective randomized double-blinded cross-over trial (MULTIWAVE study) | Trials
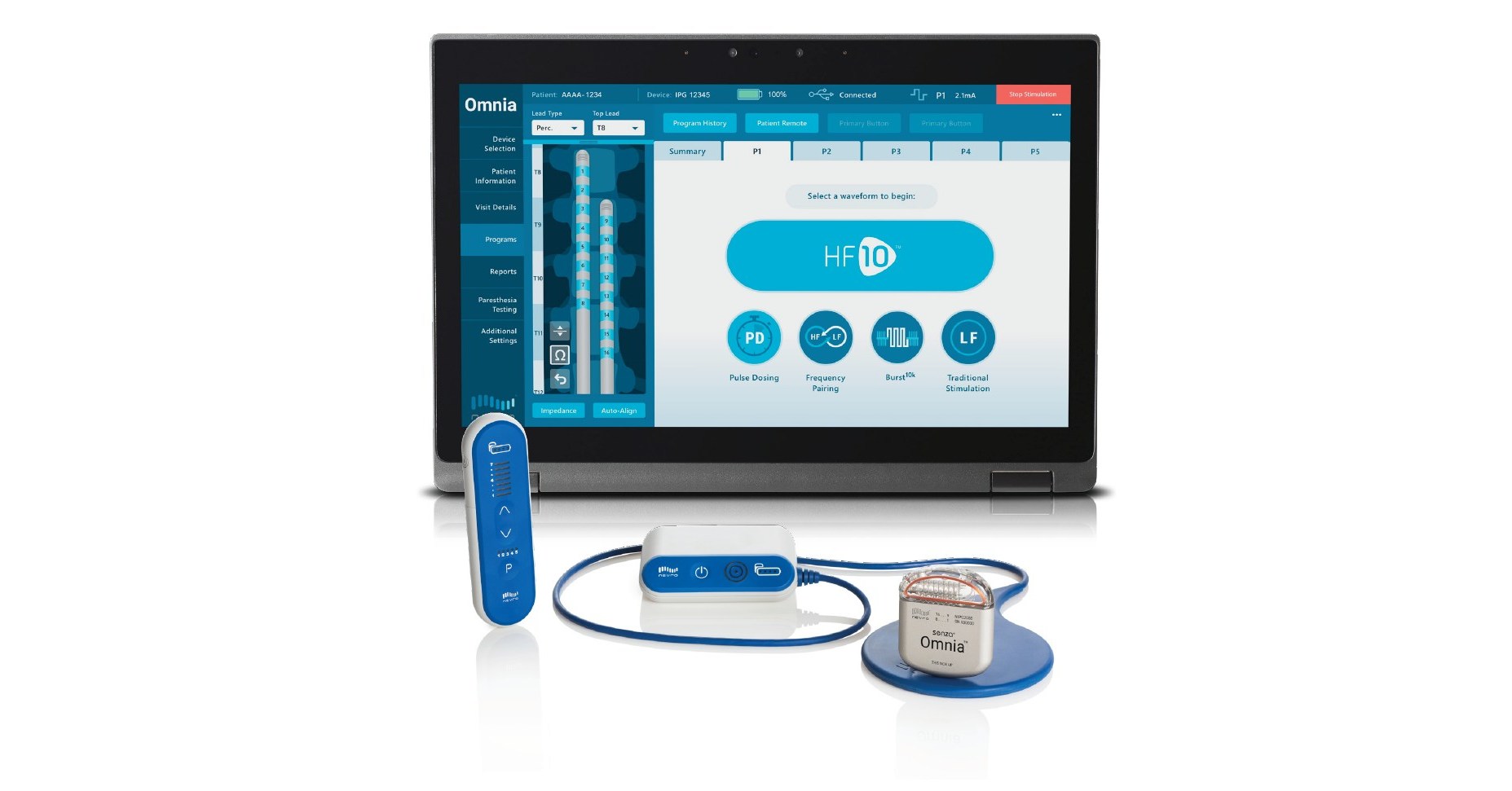
Nevro Announces CE Mark Approval of Senza® Omnia™ Spinal Cord Stimulation System to Treat Chronic Pain

Nevro Receives FDA Approval For Senza II Spinal Cord Stimulation System Delivering HF10 Therapy | Orthopedic Design Technology