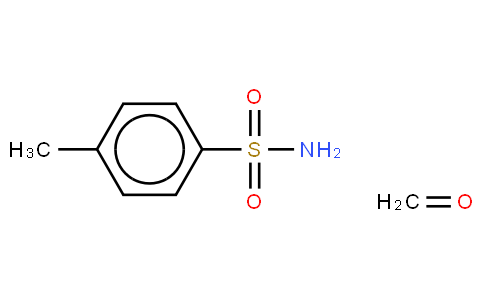
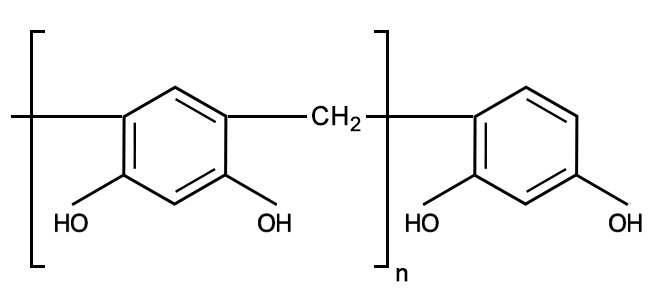
Synthesis of multifunctional epoxy resin of bisphenol-C-formaldehyde (EBCF) | Download Scientific Diagram

Adsorption of Lead Ions from Aqueous Solution by Functionalized Polymer Aniline–Formaldehyde Condensate, Coated on Various Support Materials | SpringerLink

Three-Dimensional Interconnected Microporous Carbon Network Derived from Aniline Formaldehyde Resin/Sodium Polyacrylate Interpenetrating Polymer Networks (AF/PAAS IPNs) with Controllable Porosity for Supercapacitors | ACS Applied Energy Materials

PDF) Synthesis and Thermal and Textural Characterization of Aniline Formaldehyde-Organoclay Composites
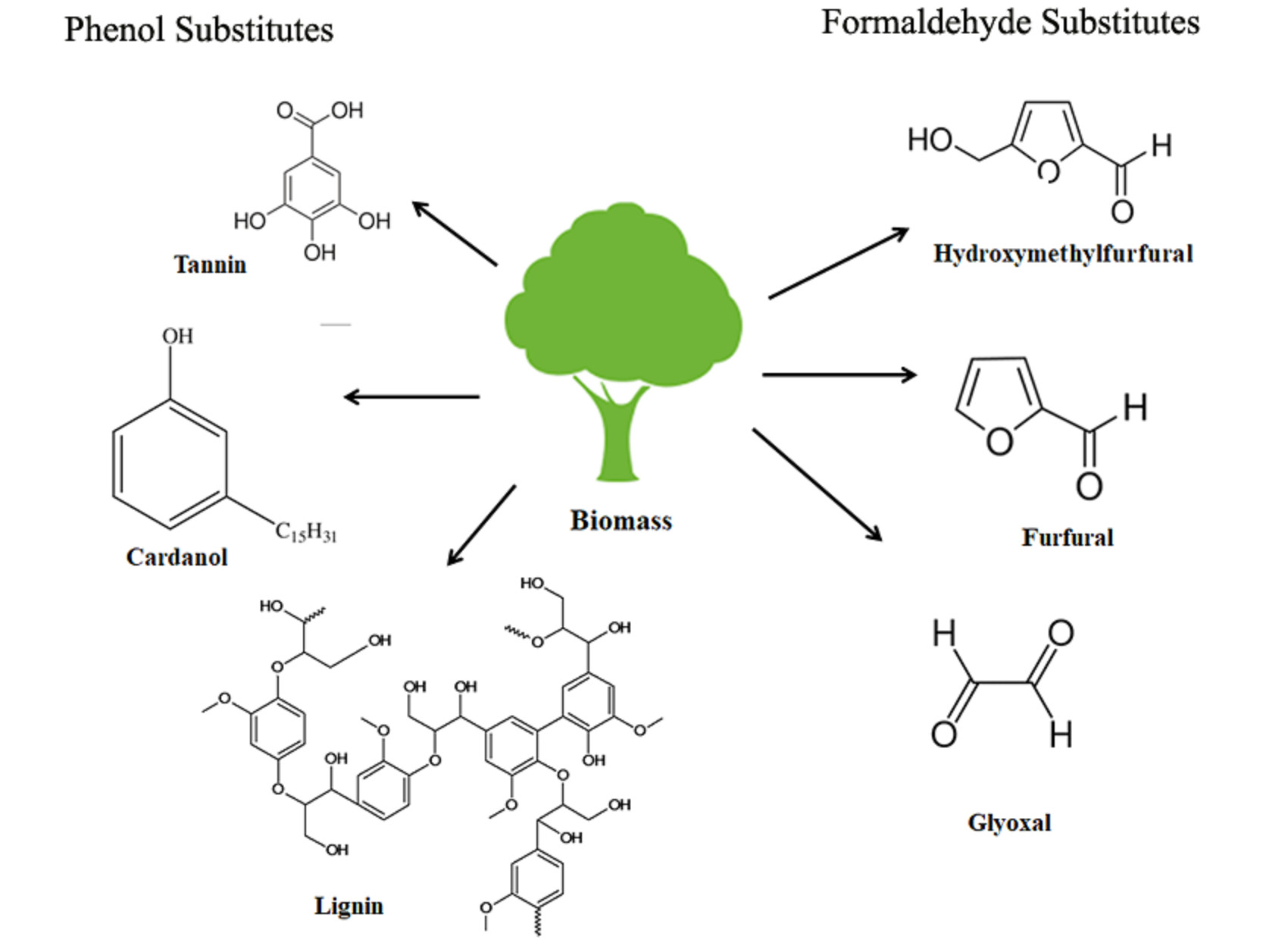
Polymers | Free Full-Text | Bio-Based Alternatives to Phenol and Formaldehyde for the Production of Resins

Reaction scheme detailing aniline polycondensation with formaldehyde... | Download Scientific Diagram

Synthesis and characterization of cobalt nanocomposite using aniline-formaldehyde resin - ScienceDirect
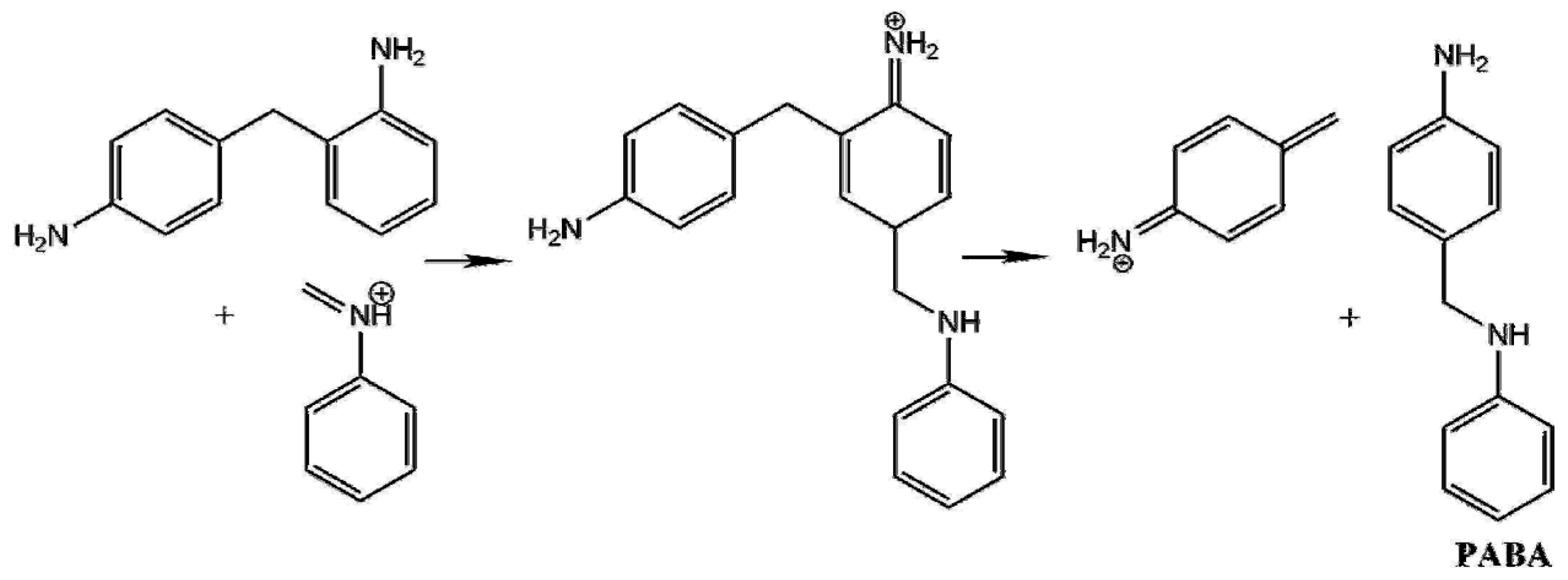
Polymers | Free Full-Text | An Ab Initio Investigation of the 4,4′-Methlylene Diphenyl Diamine (4,4′-MDA) Formation from the Reaction of Aniline with Formaldehyde

A review of recent progress in melamine-formaldehyde resin based nanocomposites as coating materials - ScienceDirect

In Situ Generation of Ammonia: an Efficient Catalyst for the Synthesis of Phenol-Aniline-Formaldehyde Resol

Effect of aniline formaldehyde resin on the conjugation length and structure of doped polyaniline: Spectral studies - Ho - 2005 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library
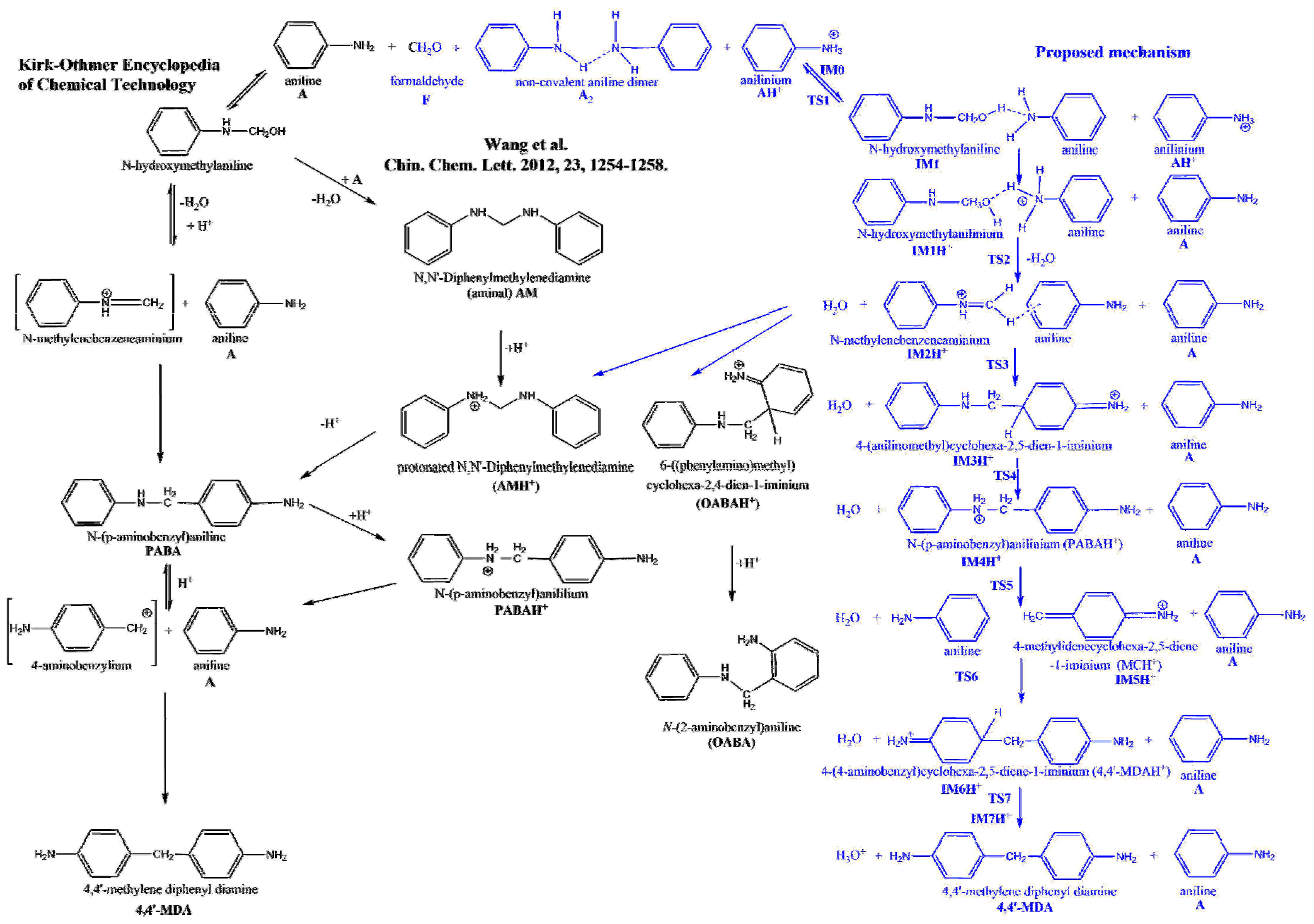
Polymers | Free Full-Text | An Ab Initio Investigation of the 4,4′-Methlylene Diphenyl Diamine (4,4′-MDA) Formation from the Reaction of Aniline with Formaldehyde
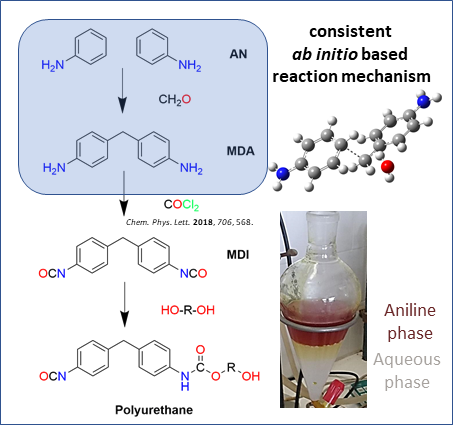
Polymers | Free Full-Text | An Ab Initio Investigation of the 4,4′-Methlylene Diphenyl Diamine (4,4′-MDA) Formation from the Reaction of Aniline with Formaldehyde

Nanostructured Aniline Formaldehyde Resin/Polysilazane Hybrid Materials by Twin Polymerization - Kaßner - 2016 - Macromolecular Chemistry and Physics - Wiley Online Library